When a gas is compressed, It increase the pressure since the volume of gas is decreased according to the gas laws, which increases the randomness of the gas molecules which start to collide more frequently and hence the temperature rises. Thus, due to compressing gas both pressure and temperature increases.
This same reason causes compressed air to feel cold as it expands and draws heat from its surroundings. The liquid which is sprayed while the can is upside-down absorbs more heat as it changes from a liquid to a gas when sprayed.
Decreasing Pressure
The combined gas law states that the pressure of a gas is inversely related to the volume and directly related to the temperature. If temperature is held constant, the equation is reduced to Boyle's law. Therefore, if you decrease the pressure of a fixed amount of gas, its volume will increase.Air heats up mainly by being in contact with hot objects. The sun shines through the air and heats it very little, but if you have a hot mesh the air passing through it will heat up. The sun heats the ground and the air in contact with the ground heats up.
The atoms and molecules in gases are much more spread out than in solids or liquids. They vibrate and move freely at high speeds. Gas can be compressed much more easily than a liquid or solid. (Think about a diving tank – 600 L of gas is compressed into a 3 L cylinder.)
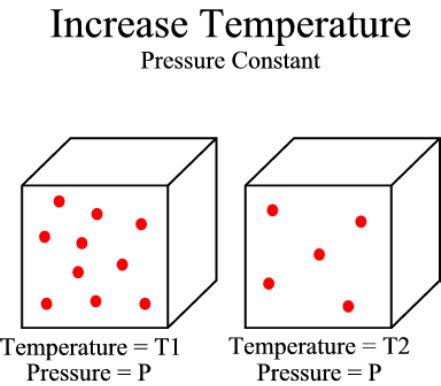
Charles' law (Gay-Lussac's law)
This law states that the volume and temperature of a gas have a direct relationship: As temperature increases, volume increases, when pressure is held constant. Heating a gas increases the kinetic energy of the particles, causing the gas to expand.Pressure by itself does not produce heat. An increase in pressure does produce heat. As you compress a quantity of gas, for instance, by pushing a piston into a cylinder, you cause every molecule that bounces into the piston wall to bounce back a little faster. An increase in pressure does produce heat.
The volume of the gas increases as the temperature increases. As temperature increases, the molecules of the gas have more kinetic energy. They strike the surface of the container with more force. If the container can expand, then the volume increases until the pressure returns to its original value.
The gas laws consist of three primary laws: Charles' Law, Boyle's Law and Avogadro's Law (all of which will later combine into the General Gas Equation and Ideal Gas Law).
When you heat a gas, both its vapor pressure and the volume it occupies increase. The individual gas particles become more energetic and the temperature of the gas increases. At high temperatures, the gas turns into a plasma.
Decreasing Pressure
The combined gas law states that the pressure of a gas is inversely related to the volume and directly related to the temperature. If temperature is held constant, the equation is reduced to Boyle's law. Therefore, if you decrease the pressure of a fixed amount of gas, its volume will increase.Charles' Law: The Temperature-Volume Law. This law states that the volume of a given amount of gas held at constant pressure is directly proportional to the Kelvin temperature. As the volume goes up, the temperature also goes up, and vice-versa.
If a gas is heated, its particles move around more quickly. They hit the walls of their container harder and more often. This increases the pressure. Sometimes the pressure gets so great that the container bursts.
The Gas Laws: Pressure Volume Temperature Relationships
- Boyle's Law: The Pressure-Volume Law.
- Charles' Law: The Temperature-Volume Law.
- Gay-Lussac's Law: The Pressure Temperature Law.
- The Combined Gas Law.
Three Ways to Increase the Pressure of a Gas
- Increase the amount of gas. This is represented by the "n" in the equation.
- Increase the temperature of the gas. This is represented by "T" in the equation.
- Decrease the volume of the gas. This is the "V" in the equation.
Similarly, when you cool a gas (reduce T) at a fixed pressure, its volume decreases – it compresses. If you compress a gas without constraining the temperature or pressure, the ratio of temperature to pressure has to decrease.
By the above, we can say that when volume decreases, density increases. Therefore, when a gas is compressed its density increases.
The point is,in a vacuum,there is nothing that the gas can do work on! Also,there is nothing that the gas has to overcome its pressure to expand,so the work it does in expanding is zero. Using the ideal gas equation is justified because in a vacuum,the only force on the particles is the force by other ones.
It's not actually possible to give a yes or no answer. If you are thinking of a gas like water vapour then yes, if you compress it, it will become more dense and eventually its density will be the same as that of liquid water it will have essentially turned into a liquid.
When an ideal gas is compressed in a closed container, its internal energy U increases. Compressing an ideal gas increases its temperature and its internal energy increases since U = f(t) for an ideal gas.
When air is compressed rapidly, temperature increases because temperature and volume of gas are inversely proportional to each other according to gas laws. When air is compressed rapidly, its volume decreases leading to increase in temperature.
Most engines run at between 195F and 220F but the fuel ignites at any temperature that is conducive to the fuel's flash point. The range is 90F to 220F. Compression ignites the fuel in a diesel engine and the temperature of the cylinder when the fuel is introduced needs to be stable to achieve a clean burn.
Since the particles of an ideal gas have no volume, a gas should be able to be condensed to a volume of zero. As kinetic energy decreases as a gas is cooled, the particles will eventually move slowly enough that there attractive forces cause them to condense.
Yes, heat can be added to a substance without causing the temperature of the substance to rise. Since the added heat does not increase the temperature of the sample results there is no increase of kinetic energy of the molecules.