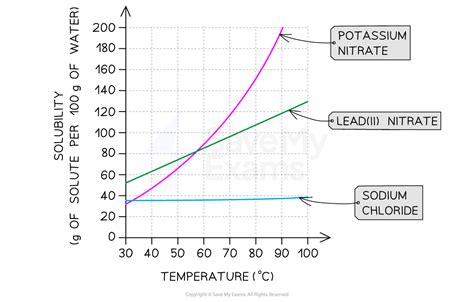
Task 1: Producing a graph to represent solubility data
| Potassium nitrate (KNO3) | Sodium nitrate (NaNO3) |
|---|
| Temp. °C | g / 100 g water | g / 100 g water |
|---|
| 30 | 45 | 96 |
| 40 | 63 | 105 |
| 50 | 84 | 114 |
For many solids dissolved in liquid water, the solubility increases with temperature. The increase in kinetic energy that comes with higher temperatures allows the solvent molecules to more effectively break apart the solute molecules that are held together by intermolecular attractions.
Sodium nitrate is a white deliquescent solid very
soluble in
water.
Sodium nitrate.
| Names |
|---|
| Boiling point | 380 °C (716 °F; 653 K) decomposes |
| Solubility in water | 73 g/100 mL (0 °C) 91.2 g/100 mL (25 °C) 180 g/100 mL (100 °C) |
The solubility of KNO3 in water increases with the increase in temperature since dissolution of KNO3 in water is an endothermic process. The solubility of CaSO4 in water decreases with the increase in temperature since dissolution of CaSO4 in water is an exothermic process.
What is the melting point of sodium nitrate?
Unlike sodium oxalate, there is a large difference in sodium nitrate solubility in different background electrolytes. Sodium nitrate is 3–4 times more soluble in NaNO2 solutions than NaOH solutions at the same sodium molality (Figure ?14).
For example, when water solutions of calcium nitrate and sodium carbonate are mixed, calcium carbonate precipitates from the solution while the other product, sodium nitrate, remains dissolved. It is a precipitate.
Factors affecting solubility
- Temperature. Basically, solubility increases with temperature.
- Polarity. In most cases solutes dissolve in solvents that have a similar polarity.
- Pressure. Solid and liquid solutes.
- Molecular size.
- Stirring increases the speed of dissolving.
This means just slightly more energy must be put into the solution than is released back into the solution; therefore dissolving table salt in water is endothermic. More energy is released into the solution than is required to pull apart the ions; therefore dissolving sodium hydroxide in water is exothermic.
If we heat the solvent, the average kinetic energies of its molecules increases. Hence, the solvent is able to dislodge more particles from the surface of the solute. Thus, increasing the temperature increases the solubilities of substances. For example, sugar and salt are more soluble in water at higher temperatures.
The solubility is a measure of the concentration of the dissolved gas particles in the liquid and is a function of the gas pressure. As you increase the pressure of a gas, the collision frequency increases and thus the solubility goes up, as you decrease the pressure, the solubility goes down..
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come into contact with the sugar more often, causing it to dissolve faster.
The solubility of a solid may increase or decrease with increasing temperature, whereas the solubility of a gas decreases with an increase in temperature and a decrease in pressure.
The heat of solution of NaCl is very small as the heat of ionization is nearly equal to the heat of hydration. Therefore, temperature doesn't affect the dissolution of NaCl as the heat of hydration almost equalizes heat of dissolution.
A saturated solution is a solution in which no more of a solute can be dissolved in a solvent.So to dissolve more of the solute we can heat the solution. Thus the increase in temperature(heat) causes more of the solute to dissolve in the molecules of the solvent.
A: The rate of dissolving is influenced by several factors, including stirring, temperature of solvent, and size of solute particles.
Increasing the temperature will therefore increase the solubility of the solute. An example of a solute whose solubility increases with greater temperature is ammonium nitrate, which can be used in first-aid cold packs. Ammonium nitrate dissolving in solution is an endothermic reaction.
The solubility of a substance in water increases on increasing the temperature. Larger amount of a substance can be dissolved in a given amount of water on heating it. The solubility of a substance decreases on lowering the temperature.
Increasing the temperature increases the rate of dissolving because, at higher temperatures, the solvent molecules are moving more rapidly and therefore come into contact with and solvate the solute molecules more rapidly.
CASE II: Increase in solubility with temperature:The addition of more heat facilitates the dissolving reaction by providing energy to break bonds in the solid. This is the most common situation where an increase in temperature produces an increase in solubility for solids.
Solubility is the maximum amount of a substance that will dissolve in a given amount of solvent at a specific temperature. There are two direct factors that affect solubility: temperature and pressure. Temperature affects the solubility of both solids and gases, but pressure only affects the solubility of gases.
Definition of SolubilityThe solubility of a substance fundamentally depends on the solvent used, as well as temperature and pressure. The solubility of a substance in a particular solvent is measured by the concentration of the saturated solution.
Trace the solubility of a substance with increasing temperature.
- The curved line represents saturation.
- Below the curve, the solution is unsaturated.
- Above the curve the solution is supersaturated. This means there is more solute than the solution can hold.
What are the customary units of solubility on solubility curves? Degress Celsius and grams of solute/100g of water 2. Define solubility. A measure of how much solute can dissolve in a given amount of solvent.
Answer. The solubility of common salt increases with rapid increase in temperature. Yes it is TRUE because due to increase in the temperature particles of water possesses high kinetic energy and move rapidly due to which space is get created for salt particles to get in the water.
Solubility at 293 K(d) The solubility of most of the salts increases in with increase in temperature. But for same salts like sodium chloride there is no effect of temperature on the solubility. For same salts like Cerium sulphate the solubility decreases with increase in temperature.